The year 2024 opens with good prospects for the treatment of antibiotic-resistant bacterial infections and the future of drug research.
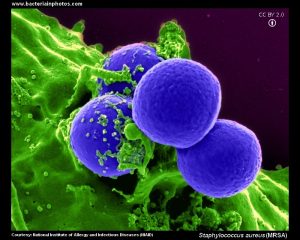
A new class of antibiotics has been discovered that, both in laboratory and mouse experiments, has proved effective against strains of Staphylococcus aureus methicillin-resistant, the dreaded MRSA (Methicillin Resistant Staphylococcus aureus).
Infections caused by MRSA bacterial strains cause more than 100,000 deaths worldwide every year, so this news has raised high expectations and hopes. Consider also that no new antibiotics have been approved in the last 50 years against Gram-negative bacteria, which have thin cell walls, whereas MRSA are Gram-positive bacteria, which also have thicker cell walls.
 The discovery of this new class of antibiotics is the result of a study, published in the journal Nature, and is due to researchers at MIT (Massachusetts Institute of Technology), who used Artificial Intelligence (AI) algorithms and in particular 'deep learning' to sift through some 12 million compounds in search of new molecules.
The discovery of this new class of antibiotics is the result of a study, published in the journal Nature, and is due to researchers at MIT (Massachusetts Institute of Technology), who used Artificial Intelligence (AI) algorithms and in particular 'deep learning' to sift through some 12 million compounds in search of new molecules.
One extremely interesting aspect is that the researchers also managed to identify what information the AI was looking for, and this could prove crucial for designing other drugs that are even more effective than those identified so far. To achieve their goal, the scientists modified an existing search algorithm, the 'Monte Carlo tree search', to generate not only an estimate of the antibacterial activity of each molecule, but also a prediction of which part of the compound is responsible for that property.
To further narrow down the candidate drugs, the researchers refined the search by training an additional three deep learning models to predict which compounds were toxic to three different types of human cells, in order to identify molecules that could kill microbes with minimal adverse effects on the human body. The researchers thus selected around 280 molecules to test in the laboratory: two of these, belonging to the same class, were particularly effective against MRSA
Experimental work revealed that the compounds appeared to kill the bacteria by altering their ability to maintain the Transmembrane Electrochemical Proton Gradient (gpet), a fundamental vital mechanism active in the mitochondria that allows electrochemical potential to be generated within the electron transport chain. The potential energy of protons produces the final synthesis of ATP, the molecules that cells use to store energy.
Experimental evidence shows that the new class of molecules can be active against Gram-positive pathogens by selectively dissipating the proton motive force in bacteria. Moreover, the new molecules selectively attack cell membranes, so that they do not cause substantial damage to human cell membranes.
This research model, which is based on the identification of substructures to design chemical compounds from scratch, opens up exciting prospects for researchers who will also be able to make use of the 'out of the box' approach, which has made it possible to identify the algorithms used by AI in the selection process of new molecules.
Read also our articles on antibiotic therapy:
- New antifungals also in the pipeline for ophthalmology? - Italian Ophthalmologist
- Antibiotics and cytotoxicity - Oculista Italiano
- Antibiotic resistance in eye infections - Oculista Italiano
- Pharmacists and antibiotic resistance - Oculista Italiano
Wong F, Zheng EJ, Valeri JA, Donghia NM, Anahtar MN, Omori S, Li A, Cubillos-Ruiz A, Krishnan A, Jin W, Manson AL, Friedrichs J, Helbig R, Hajian B, Fiejtek DK, Wagner FF, Soutter HH, Earl AM, Stokes JM, Renner LD, Collins JJ. Discovery of a structural class of antibiotics with explainable deep learning. Nature. 2023 Dec 20. doi: 10.1038/s41586-023-06887-8. Epub ahead of print. PMID: 38123686.
