We republish the interview with Dr Nino Asero that accompanied the birth of the project that led to the new drug for the medical treatment of Acanthamoeba keratitis
Dr. Asero, could you summarise the characteristics of this type of keratitis?
Acanthamoeba keratitis (AC) is a rare, severe and seriously debilitating corneal infection caused by Acanthamoeba spp, a ubiquitous protozoan present in soil, air and water1. Approximately 85% of CA cases occur in contact lens wearers, due to improper lens wear, although the disease can occur after corneal trauma, especially in rural settings. Although it has a low incidence (1 in 100,000 in Europe), CA is a potentially devastating eye infection for patients, as it can cause intense pain, vision deterioration and, if not effectively treated, enucleation and blindness.
Why is the pathology so devastating?
This damaging development is due to the infectious agent: theAcanthamoeba spp is a protozoan whose life cycle includes a dormant cyst stage and an infectious stage as an active motile trophozoite. Cysting allows the micro-organism to survive in adverse environmental conditions. In particular, it is the dormant stage that is the basis for the persistence of the infection.
What are the clinical signs and symptoms of this disease?
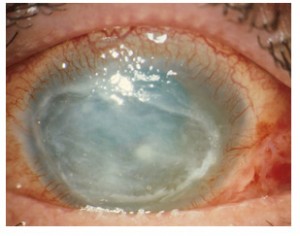
As for the clinical signs, CA can result in epithelial microerosions that may evolve into irregular anterior stromal infiltrates, corneal ulcers, melting and dense corneal abscesses in advanced stages of the disease. Typically, perineural involvement is characterised by severe ocular pain.
I symptoms are generally similar to those of other eye infections (pain, redness, irritation, blurred vision, hypersensitivity to light and excessive tearing) and may persist for several weeks.
Early diagnosis certainly improves outcome2 of infection, which, if undiagnosed, may progress for 4-8 weeks, resulting in an anterior infiltrate that may remain in the central cornea or give rise to a classic ring-shaped abscess, accompanied by limbitis, episcleritis and occasionally scleritis. Epithelial scraping can detect the amoeba. However, if it fails, the infection progresses to a large, deep infiltrated ulcer.
What is the current therapeutic approach used to manage this form of keratitis?
Effective therapy must be based on drugs that can suppress the cyst in both phases of its life cycle in order to completely eradicate the disease.
In fact, there is currently no authorised drug for the treatment of CA in any country. However, in recent years, the administration of (off-label) combinations of anti-amœbic agents, such as biguanides and diamidines, has greatly improved outcomes. The use of biguanides, in particular, has revolutionised the course of CA, especially when treated in the early stages, as pain relief is rapid and the active infection is usually eradicated after 4 weeks, compared to 4 months or more with propamidine and neomycin therapy.
Biguanide polyhexanide (PHMB) and chlorhexidine are in fact the only active ingredients effective in the cystic phase of the protozoa.
The treatment currently used involves the administration of a biguanide (PHMB 0.02% or Chlorhexidine 0.02%) and a diamidine, although no clinical data are available to show that the combination is more effective than PHMB monotherapy.
This therapy gave good results in most patients (90%)3 and, started early, allows full visual recovery. However, the dosage has not been standardised and a meaningful evaluation of safety and efficacy is lacking.4 according to the standard criteria laid down in European and international drug development guidelines.
How does the ODAK project fit into this context?
ODAK (Orphan Drug for Acanthamoeba Keratitis) is a European research project for the development of a stable and safe formulation of PHMB for the effective pharmacological treatment of Acanthamoeba Keratitis. PHMB has already received from the EMA (European Medicines Agency), at the request of SIFI, the designation of Orphan Drug (Orphan Drug) for the treatment of CA.
The ODAK project, of which SIFI is leader, involves several Partners (see box) with complementary experience and specific expertise in the clinical management of acanthamoeba keratitis. The project is co-funded by the FP7 programme of the European Commission and will last a total of 60 months.

Research and development activities include trials in pharmaceutical technology and pharmacokinetics/pharmacodynamics and toxicology, clinical studies and an industrial development phase of the final product.
In what terms could centres specialised in infectious eye diseases be involved?
Non-clinical development studies are currently underway to identify the most stable and safe formulation. These will last a total of about 24 months, to be followed by the clinical phase of safety and efficacy in humans. Crucial to the development of the clinical programme is a retrospective study conducted at two leading clinical centres, Moorfields Eye Hospital in London (Prof. John Dart) and S. Raffaele in Milan (Dr. Paolo Rama).
Particularly in clinical efficacy trials it will be necessary to recruit a sufficiently adequate number of patients that is statistically significant. It is easy to see how, since Acanthamoeba keratitis is a rare disease, the involvement of research centres that are more involved in recruiting patients with this disease will be appropriate and desirable at this stage in order to quickly reach the desired goal.
In this regard, if your readers wish, they can obtain further information by visiting the project website www.odak-project.eu or write to clinicians@odak-project.eu.
Bibliography
1. Schuster FL, Visvesvara GS. Opportunistic amoebae: challenges in prophylaxis and treatment. Drug Resist Updat. 2004 Feb;7(1):41-51.
2. Claerhout I, Goegebuer A, Van Den Broecke C, et al. Delay in diagnosis and outcome of Acanthamoeba keratitis. Graefes Arch Clin Exp Ophthalmol. 2004 Aug;242(8):648-53.
3. Seal D. Treatment of Acanthamoeba keratitis. Expert Rev Anti Infect Ther. 2003 Aug;1(2):205-8.
4. Dart JK, Saw VP, Kilvington S. Acanthamoeba keratitis: diagnosis and treatment update 2009. Am J Ophthalmol. 2009 Oct;148(4):487-499.
